Don't hesitate to send a message
The design and production comply with ISO8537. The plastic parts are moulded by ...
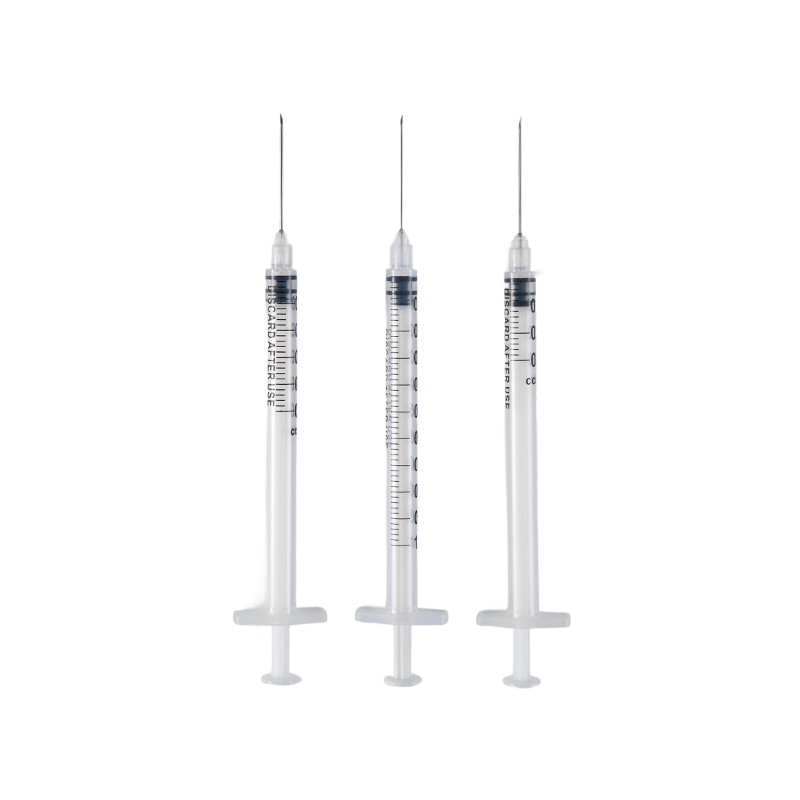
A Disposable Syringe plays a central role in daily medical procedures. From vaccination campaigns to routine injections, healthcare facilities rely on consistent performance and reliable structure. Smooth plunger movement, clear scale markings, and stable sealing are not luxury features—they are operational requirements.
Clinicians expect accurate dosage delivery without hesitation during administration. That expectation directly relates to how the syringe is designed and manufactured. A stable barrel, precise piston fit, and proper lubrication together create predictable injection performance.
The barrel of a Disposable Syringe is made from highly transparent polypropylene (PP). This material provides a bright and clear appearance, allowing medical staff to observe the liquid condition and air bubbles easily. Visual clarity reduces dosing errors and supports confident operation.
Polypropylene also offers structural stability. It maintains its shape under pressure and supports precise graduation printing. When scale markings remain sharp and readable, dosing accuracy improves during high-volume procedures.
Inside the barrel, the piston plays an equally important role. Two material options are available: synthetic rubber and natural rubber. Each option offers distinct tactile feedback and elasticity. The tolerance between barrel and piston is carefully controlled to maintain smooth sliding performance without looseness.
Medical-grade silicon is applied as lubrication. This supports consistent plunger motion and reliable sealing during injection. The combination of material compatibility and lubrication design directly influences user experience.
Modern Disposable Syringe Suppliers invest heavily in automation. The component mold injection and assembly processes are performed by fully automatic machines. This approach reduces direct human contact during production, lowering the chance of contamination from manual handling.
Automation also improves dimensional consistency. When injection molding parameters remain stable, the barrel thickness, nozzle geometry, and plunger alignment maintain uniformity across production runs. That uniformity translates into a stable injection force during clinical use.
Production environments operate under strict control procedures. Monitoring systems track molding temperature, cycle time, and assembly precision. Quality checks are conducted at multiple stages, including appearance inspection, leakage testing, and functional sliding evaluation.
For hospitals and distributors, controlled production provides supply reliability. When working with experienced Disposable Syringe Suppliers, batch traceability and consistent documentation support regulatory compliance and internal audits.
Packaging format affects storage efficiency and handling convenience. Disposable Syringe Suppliers typically provide two main packing methods:
Individual blister packaging supports sterile barrier protection for single-unit distribution. It is suitable for hospital wards, clinics, and pharmacies where one-by-one dispensing is required.
Bulk packaging reduces packaging material and supports high-volume procurement models. It is commonly selected for centralized sterilization workflows or institutional redistribution systems.
Packaging decisions often depend on logistical planning rather than product structure. Understanding end-use workflow helps determine which option aligns with operational needs.
A Disposable Syringe may appear simple in design, yet its performance depends on material science, precision molding, and disciplined assembly processes. From piston elasticity to lubrication consistency, each component contributes to overall usability.
In medical environments where efficiency and reliability are daily requirements, structured production and consistent quality management remain central. Collaboration with capable Disposable Syringe Suppliers provides stability across procurement cycles and supports long-term distribution strategies.
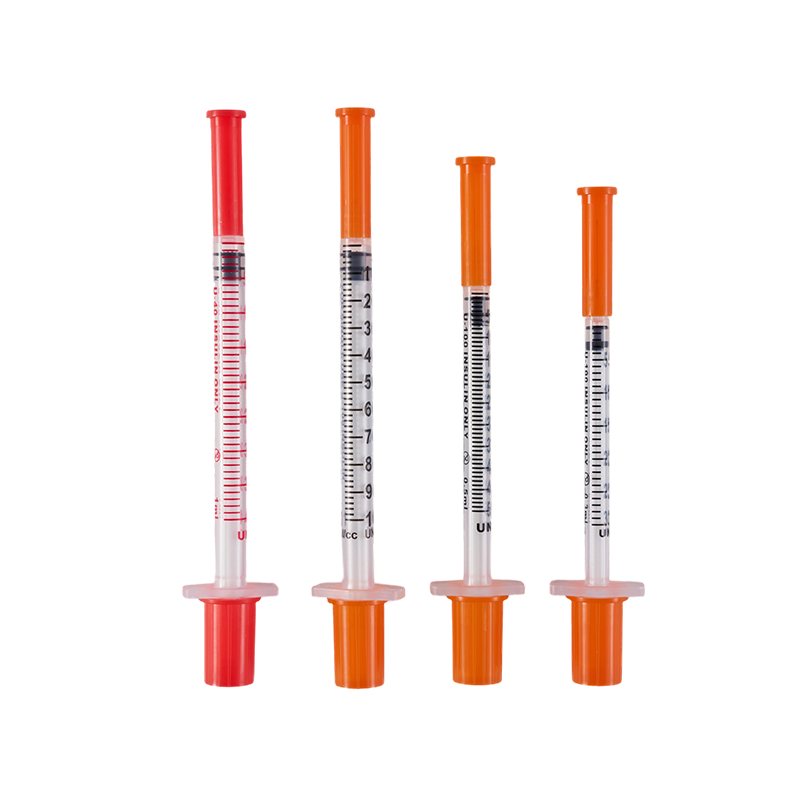
The design and production comply with ISO8537. The plastic parts are moulded by ...

There are three kinds of the tip types, Sealed-circle with 2 side holes, sealed-...

Assembling with insulin pen, for insulin hypodermic injection.The plastic parts ...

Used in conjunction with an insulin pen, it is used for subcutaneous injection o...

The cannula is made of high quality austenite stainless steel.All the components...
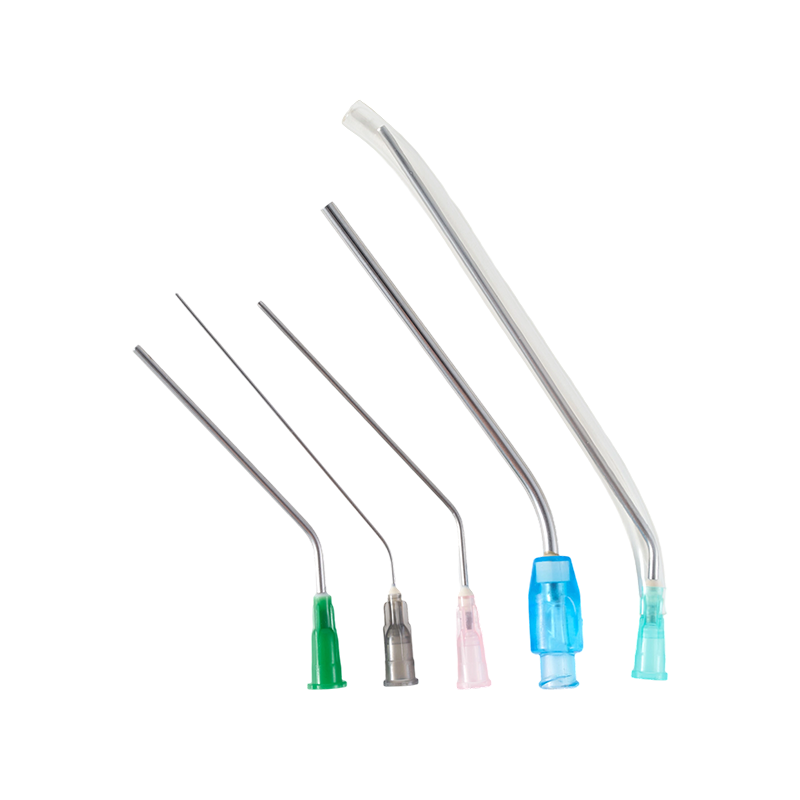
The material of the needle is Medical grade SUS304,which have great stiffness, t...

The barrel is made from high transparent polypropylene(PP),which have a bright a...

The cannula is made of high quality austenite stainless steel.The lancet tip is ...