Don't hesitate to send a message
The design and production comply with ISO8537. The plastic parts are moulded by ...
A Medical Needle is a small component in appearance, yet it carries a significant function in clinical procedures. From routine injections to vaccination programs and biopharmaceutical filling, stable puncture performance and dosage accuracy are daily operational requirements.
Healthcare professionals expect consistent penetration force, stable connection with syringes, and clear dosage alignment. When needle geometry or material quality fluctuates, injection efficiency can be affected. For institutions managing large procurement volumes, production stability and documentation transparency from Medical Needle Suppliers are part of routine evaluation.
Understanding how a needle is manufactured helps clarify why some products perform more consistently than others.
Production begins with material selection. The syringe components connected to the Medical Needle are made of medical-grade polypropylene. This material supports structural integrity and compatibility with sterilization processes.
Medical Needle Suppliers operating under ISO quality systems follow structured quality management protocols. Each batch is processed under defined parameters, from injection moulding to final assembly. Controlled environments reduce contamination risks during production.
Precision injection moulding plays a key role in forming stable hubs and connectors. Dimensional accuracy affects how securely the needle attaches to the syringe. Poor tolerance control may result in leakage or instability during injection.
Ethylene oxide sterilization is used to maintain sterile conditions before shipment. This method supports non-toxic and non-allergenic product characteristics. For healthcare facilities, consistent sterilization validation is an important part of supply evaluation.
One of the practical concerns in clinical environments is puncture smoothness. The bevel of a Medical Needle is laser-polished. This process refines the surface, reducing resistance during penetration.
A smoother bevel surface contributes to more controlled insertion. Medical staff often recognize the difference in tactile feedback when bevel finishing is uniform. Irregular polishing can create uneven penetration force.
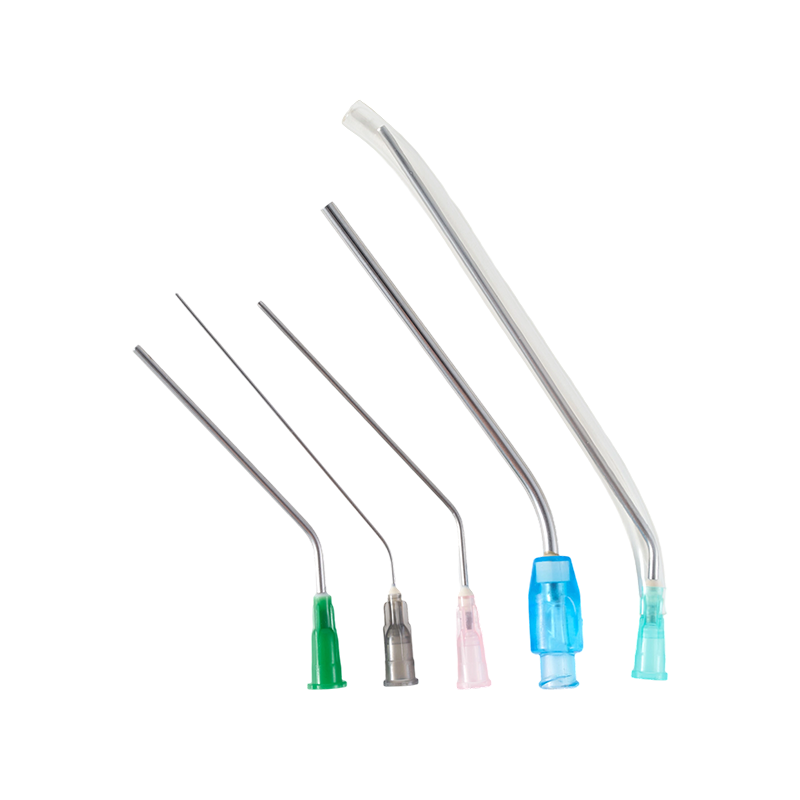
Needle diameter and length also influence application scenarios. A wide range of specifications is available to support different injection volumes and tissue types. Selection should align with clinical procedure requirements rather than general preference.
Medical Needle Suppliers typically provide compatibility options across 0.1ml to 60ml syringe systems. Clear dosage scales on matched syringes support accurate drug delivery in vaccination and therapeutic settings.
Sterility maintenance does not end at the production line. After ethylene oxide sterilization, packaging becomes a protective barrier.
Triple-sealed packaging is commonly used to reduce contamination risks during transportation and storage. Each layer functions as a physical barrier against environmental exposure. Stable sealing quality also supports long-distance logistics without compromising product integrity.
Transportation conditions can vary significantly. Temperature fluctuations and handling pressure may impact packaging stability. Structured packaging design reduces deformation risks and maintains product sterility until use.
For institutions reviewing Medical Needle Suppliers, packaging validation data and transport simulation reports are often part of the documentation review.
Stable puncture performance, reliable sterilization, and controlled packaging all contribute to dependable clinical workflows. A Medical Needle designed with laser-polished bevel surfaces and produced under controlled moulding conditions provides predictable penetration characteristics.
Ethylene oxide sterilization combined with structured packaging supports sterile distribution across hospitals, clinics, and pharmaceutical facilities. ISO-based management frameworks reinforce documentation and traceability.
In clinical environments where injection procedures are routine, device consistency simplifies operations. Cooperation with experienced Medical Needle Suppliers allows institutions to maintain structured procurement cycles supported by controlled production systems.
By focusing on material selection, moulding precision, sterilization discipline, and packaging reliability, healthcare providers can access injection components that align with daily operational requirements without unnecessary complexity.
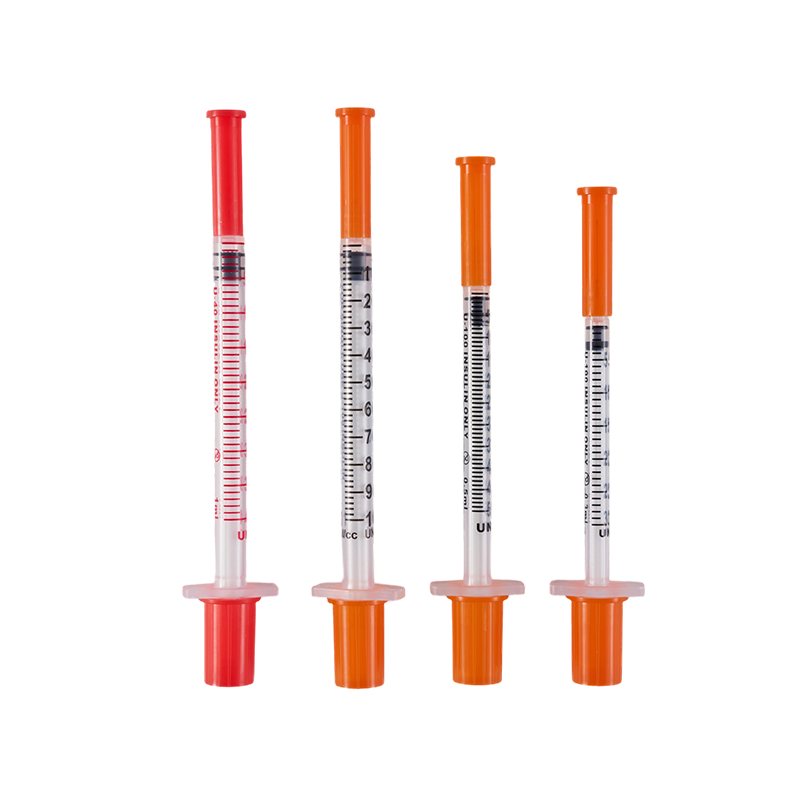
The design and production comply with ISO8537. The plastic parts are moulded by ...

There are three kinds of the tip types, Sealed-circle with 2 side holes, sealed-...

Assembling with insulin pen, for insulin hypodermic injection.The plastic parts ...

Used in conjunction with an insulin pen, it is used for subcutaneous injection o...

The cannula is made of high quality austenite stainless steel.All the components...

The material of the needle is Medical grade SUS304,which have great stiffness, t...
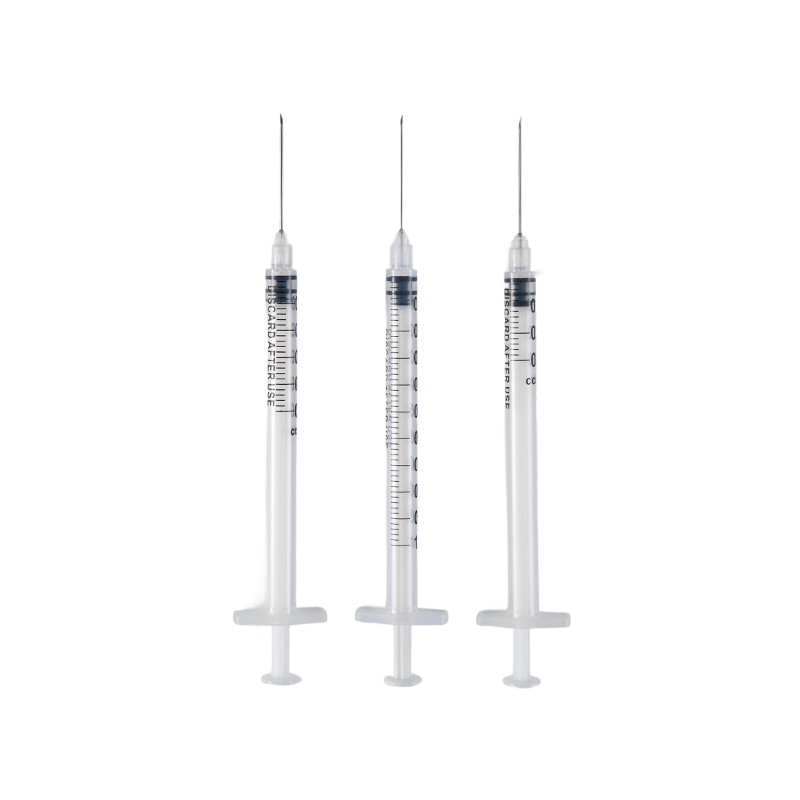
The barrel is made from high transparent polypropylene(PP),which have a bright a...

The cannula is made of high quality austenite stainless steel.The lancet tip is ...