Don't hesitate to send a message
The design and production comply with ISO8537. The plastic parts are moulded by ...
A Hypodermic Needle is a routine yet critical medical component used in hospitals, clinics, laboratories, and vaccination centers. Despite its small size, its structural precision directly affects injection performance, dosage control, and workflow efficiency.
Medical professionals expect consistent penetration force, stable hub connection, and predictable fluid flow. When needle dimensions vary or surface finishing is inconsistent, injection resistance can fluctuate. This creates inconvenience during repetitive procedures and may disrupt clinical rhythm.
The performance of a Hypodermic Needle begins with raw material choice. Medical-grade stainless steel is typically used for the cannula, offering corrosion resistance and mechanical strength. Stable material composition supports reliable puncture performance during storage and use.
The hub component is commonly manufactured through precision injection moulding using medical-grade polymers. Dimensional accuracy at the hub-cannula junction affects leak resistance and attachment stability when connected to a syringe.
Hypodermic Needle Suppliers operating under structured quality systems monitor material certification, tensile strength, and dimensional tolerance during incoming inspection. Stable raw material verification reduces variability in downstream production.
One of the practical concerns in daily use is puncture smoothness. The bevel geometry of a Hypodermic Needle plays a decisive role in penetration behavior. Multi-facet bevel grinding combined with fine polishing reduces insertion resistance.

A uniform bevel surface allows controlled entry into tissue. Inconsistent grinding may create irregular resistance, which becomes noticeable during repeated injections. Automated grinding systems help maintain angle accuracy and surface uniformity.
Surface treatment processes also influence glide performance. Polished cannulas reduce friction during insertion and withdrawal. Consistency across production batches is closely linked to machining precision and inspection discipline.
Clinical requirements vary across departments. Different procedures require specific needle diameters and lengths. A comprehensive gauge range allows alignment with vaccination, therapeutic injection, and laboratory sampling tasks.
Thinner gauges are often selected for subcutaneous applications, while thicker gauges may be used for intramuscular injections or fluid transfer. Length selection depends on anatomical considerations and injection depth requirements.
Providing structured size options supports inventory planning. Hypodermic Needle Suppliers with a complete product series reduce the need for fragmented sourcing across multiple vendors.
Dimensional consistency across gauges also contributes to operational familiarity. Medical staff benefits from predictable performance regardless of size selection.
Sterile condition maintenance is a central element in hypodermic needle production. After assembly, products undergo sterilization processes such as ethylene oxide treatment. Controlled sterilization parameters maintain product integrity while supporting non-toxic outcomes.
Packaging design plays a protective role during transport and storage. Individual blister packaging is commonly used to isolate each unit. Sealed pouches protect against environmental exposure before use.
Hypodermic Needle Suppliers maintaining systematic sterilization records and packaging inspection reports provide added transparency in supply chain management.
A Hypodermic Needle may appear simple, yet its performance depends on material selection, machining accuracy, sterilization discipline, and packaging stability. Controlled bevel grinding and polished surfaces contribute to smooth penetration. Dimensional calibration supports a reliable syringe connection.
Sterilization management and sealed packaging maintain sterile conditions during transport. Automated production systems reinforce structural consistency and batch traceability
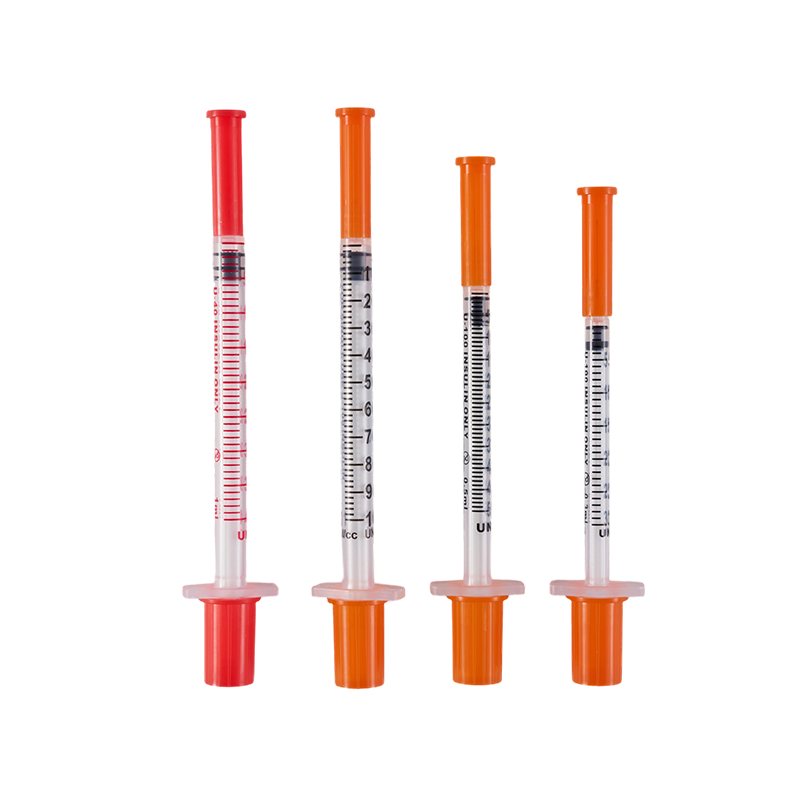
The design and production comply with ISO8537. The plastic parts are moulded by ...

There are three kinds of the tip types, Sealed-circle with 2 side holes, sealed-...

Assembling with insulin pen, for insulin hypodermic injection.The plastic parts ...

Used in conjunction with an insulin pen, it is used for subcutaneous injection o...

The cannula is made of high quality austenite stainless steel.All the components...
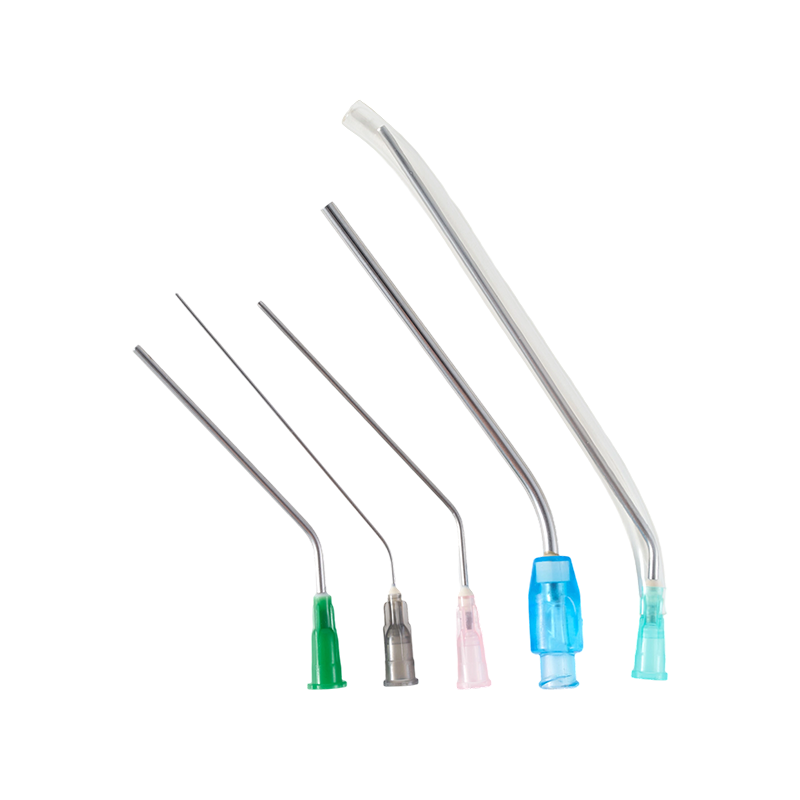
The material of the needle is Medical grade SUS304,which have great stiffness, t...
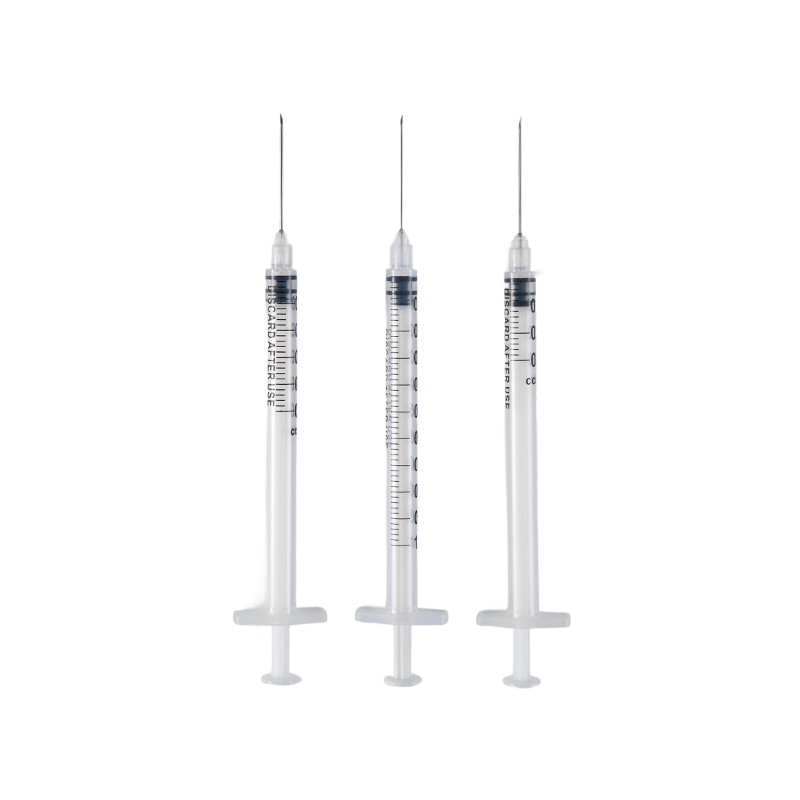
The barrel is made from high transparent polypropylene(PP),which have a bright a...

The cannula is made of high quality austenite stainless steel.The lancet tip is ...